Clinical and Translational Research
The “bench-to-bedside” (in dentistry, “bench-to-chairside”) process of harnessing knowledge from basic sciences to produce new drugs, devices, and treatments to improve clinical care to benefit human health is called Translational Research. As there are many distinct activities contributing to the overall progress of human health research, three discrete but synergistic processes have been suggested:
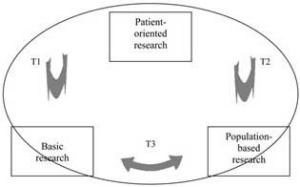
- T1 translational activities involve applying basic research findings in the lab to clinical practice via human clinical studies (trials) to ultimately change existing "standards of care."
- T2 research attempts to perform patient-oriented research that connects with population-based research focusing on improved patient outcomes, implementing best clinical practices and better overall community health
- T3 research promotes interaction between laboratory-based research and population-based research to stimulate a robust scientific understanding of human health and disease.
Other investigators have suggested a more elaborate five-phase process in translation where phase T0 focuses purely on preclinical, mechanisms-based, basic research, while T1 is further divided into two distinct phases - phase 1-2 and phase 3 (pivotal) clinical trials.


There are significant restrictions on human subject research that ensure safety is rightly given highest priority. Moreover, these clinical translational endeavors are extremely expensive with limited non-commercial avenues for support. A major barrier for clinical translational research has been a very high rate of failures to effectively applying robust basic science observations to human therapies. Besides the often cited ‘a mouse is not a man’ analogy, the lack of well-trained clinical investigators cognizant of basic science methodologies presents a formidable barrier to future progress. This has led to the description of clinical translational endeavors as a "valley of death" (Declan Butler. Nature 453, 840-842 (2008)).
Department of Oral Biology
629 BRB
Buffalo, NY 14214
Phone: 716-829-2844
Fax: 716-829-3942
Department Chair
Stefan Ruhl, DDS, PhD
shruhl@buffalo.edu
Senior Program Coordinator
Imon Candelaria, PhD
imongosw@buffalo.edu